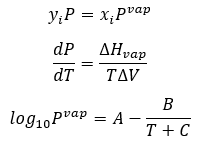Chapter 12: Equilibrium in a Multicomponent system
A multicomponent system, also known as binary mixture, can be explained by Lewis/Randall and Henry’s Law.
Lewis/Randall
Lewis/Randall
- The molar fraction of a “Xa” approaches to 1.
- The solution is almost completely a.
- Interactions a-a
- Intermolecular interaction is given by fugacity of a “fa”
- The molar fraction of a “Xa” approaches to 0.
- Interactions a-b
- a is diluted in b
- Intermolecular interaction is given by Ha
Fugacity is the tendency to escape, then the strongest interactions will have the lowest fugacity.
Vapor-Liquid Systems
The fugacity in the vapor phase is equal to the fugacity in the liquid phase.
The fugacity in the vapor phase is equal to the fugacity in the liquid phase.
In order to know more about the characteristics of the system, we consider Raoul’s Law, Clausius Clapeyron equation, and Antoine Law.
Equilibrium
In the 2 phase region, we use yi and xi, together with an overall mass balance to calculate the fraction of the overall mixture that is in vapor and in liquid. These are known as flash calculations.
For more information take a look in the next link
Applications
Analysis of multi-component systems is useful in many industrial processes. For example, knowledge of a T-x-y diagram can help set the conditions for a series of distillation columns. Another application is in separation processes. Without information about how mixtures act at various temperatures and pressures, it would be impossible to accurately design strippers or absorbers, both trayed and packed columns.
Example by thermocuates
Objective: Graph the phase diagram for the most volatile species of the substance
Problem
References
Almanza, Y. Equilibrium Thermodynamics. ITESM. 2016
Soto, M. Liquid-Vapor equilibrium. Fisicoquímica. ITESM. 2016
Soto, M. Liquid-Vapor equilibrium. Fisicoquímica. ITESM. 2016