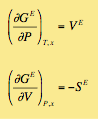Excess Gibbs Free Energy
Basis from which the activity coefficient from all species in the mixture can be obtained from a single equation.
The Excess Gbbs Free Energy Models, reduce the quantities of experimental data into few empirical parameters and provide information that can be used on different thermodynamic models.
The Excess Gbbs Free Energy Models, reduce the quantities of experimental data into few empirical parameters and provide information that can be used on different thermodynamic models.
The excess property
Are properties of solutions that exceed from the ideal solutions, at the same conditions of ideal solutions
@ ideal solutions: Excess functions = 0
@ ideal solutions: Excess functions = 0
Excess Functions: The excess functions are described just as its original fundamental functions, and partially derivated from extensive functions as shown.
Indicating E as excess condition
Indicating E as excess condition
In other way, the way that the equilibrium equation is presented as the next one:
Applications
The activity coefficient models are highly used as a tool in the engineering field to give a solution to the phase equilibria problem. This activity coefficient takes a considerable importance on the liquid-vapor equilibrium calculations. Those methods apply on constant conditions systems, at moderated pressure and temperature.